
Former Graduate Student in Chemistry
Recent molecular dynamics (MD) simulations [1] have shown that the narrow interior channel of the hydrophobic nanotube is occupied by water. Small perturbations in the interactions between water and nanotube lead to bimodal transitions between empty and filled states.
My recent work involved MD simulations of a solvated nanotube twice as long as the one described in Ref.1 (27Å long and 8.1Å in diameter).Ā Both equilibrium and nonequilibrium simualtions were performed to study the length dependence of filling and emptying kinetics. It is found that the transition time for emptying (i.e. the time to empty the tube after the transition was initiated) is significantly longer for the longer tube [2].
The sensitivity of filling with respect to variations in water-nanotube interactions was studied by tuning the carbon-water Lennard-Jones potential with a control parameter 1
that multiplies the
dispersion
term.Our results show that for l = 0.785, the system fluctuates between empty and filled states at equilibrium at 300K. The free energy of filling GN as a function of the occupation number N of water molecules in the tube is determined by the occupation probability P(N) through the relation G N /k B T =-lnP N while temperature dependence of this determines the entropy and energetics of filling. Both of these are currently under study.
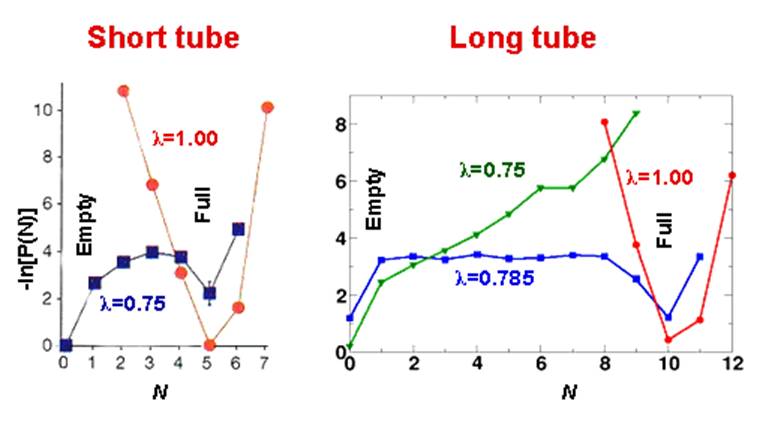 Ā
Ā
Above Fig: Free energies GN/kBT = -lnPN as a function of N for l =0.75, l=0.785 and l=1 for filling of short and long tube.
Selected Publications:
Aparna Waghe, Jayendran C. Rasaiah and Gerhard Hummer, "Entropy of single-file water in (6,6) carbon nanotubes", J.Chem.Phys, 137, 044709-9, (2012).
[2] AA.Waghe, J. C. Rasaiah and G Hummer, “ Filling and Emptying Kinetics of Carbon Nanotubes in Water”, J.Chem.Phys. 23, 10789 (2002)
